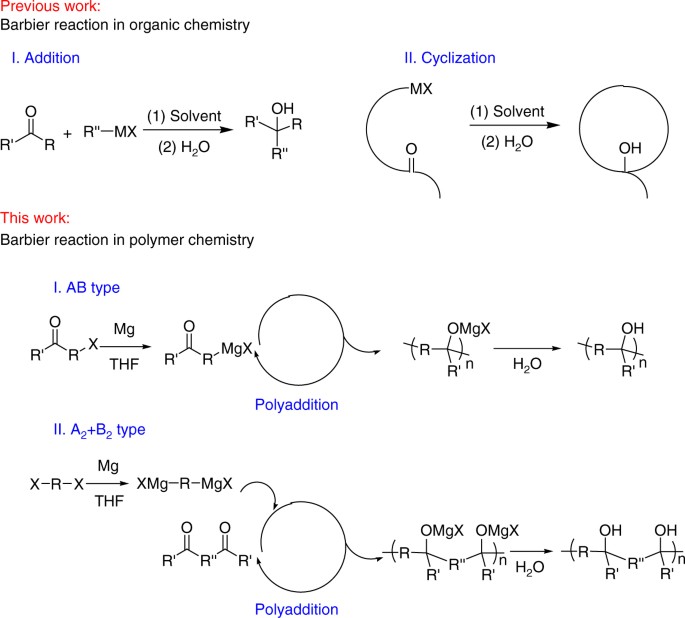
Proposed mechanism for the samarium Barbier reaction with excess HMPA. | Download Scientific Diagram
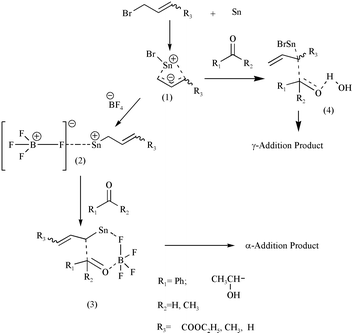
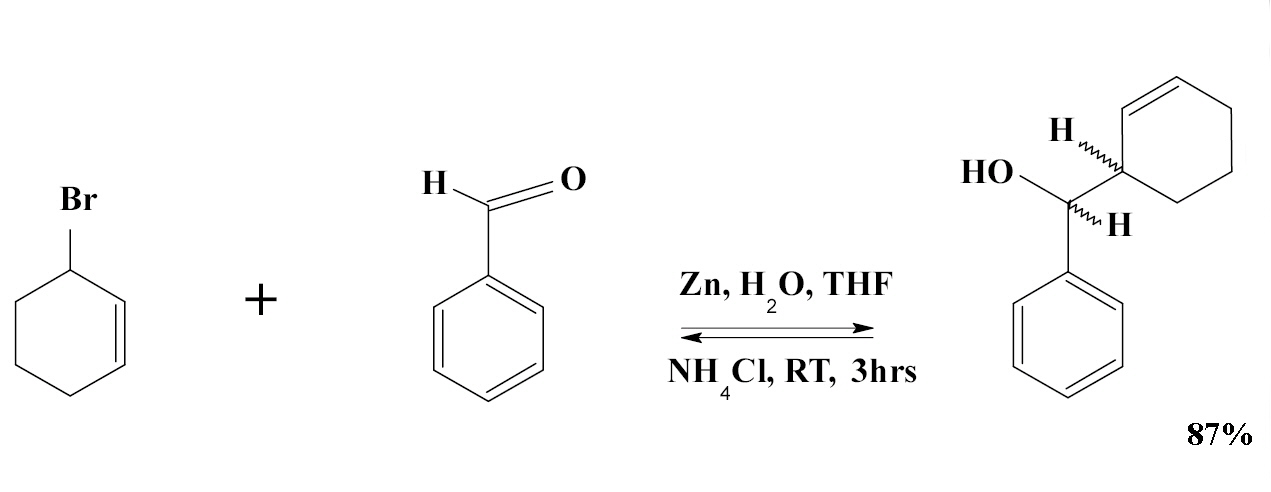
High regio- and stereoselective Barbier reaction of carbonyl compounds mediated by NaBF 4 /Zn (Sn) in water - New Journal of Chemistry (RSC Publishing) DOI:10.1039/B303187J

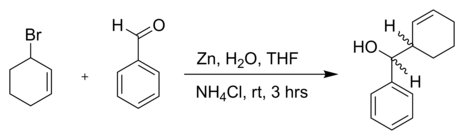
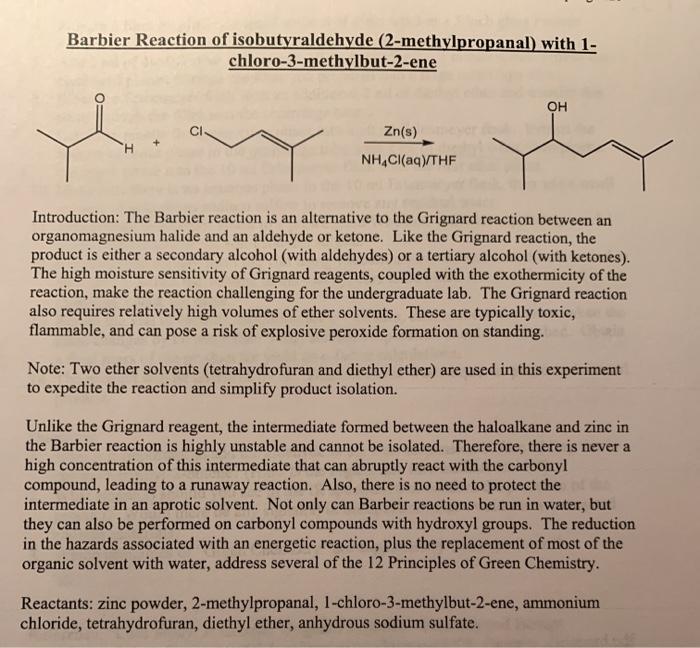
SOLVED:Znls) NH Cl(aq)THF Introduction: The Barbier reaction altemnative t0 thc Grignard reaction between organomagnesium halide and an aldehyde or ketone. Like the Grignard reaction , the product Is either secondary aleohol (with
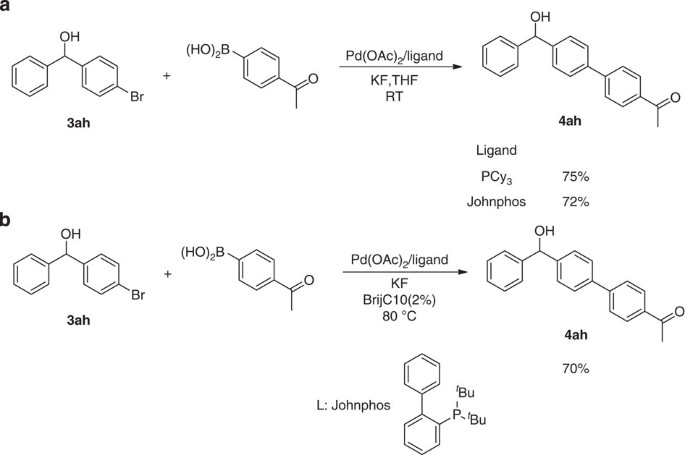
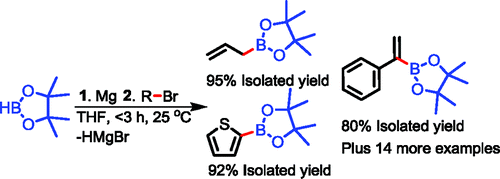
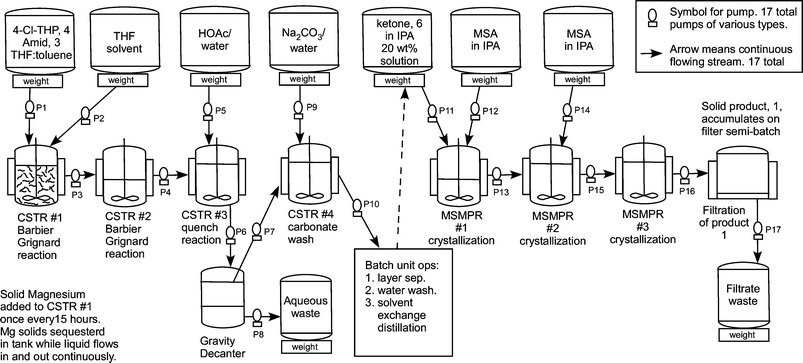
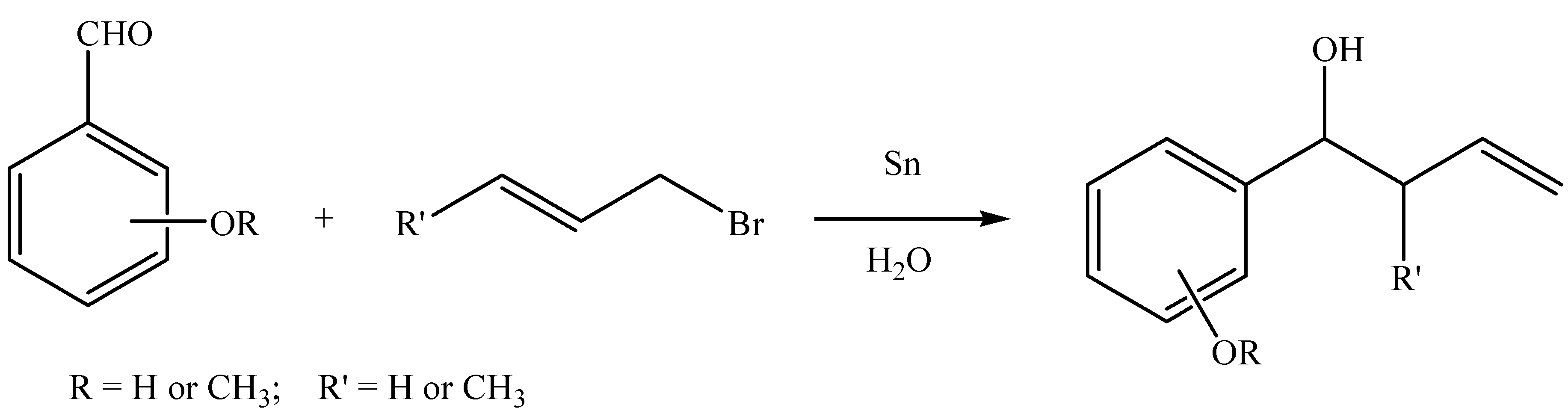
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications
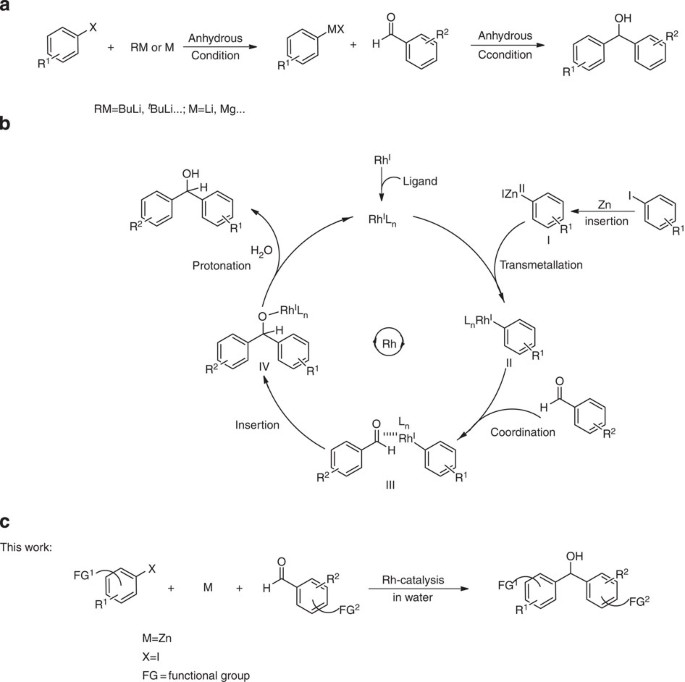
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications